About Us
Seki Aoi Techno: Your Premier Partner
in Medical Equipment Manufacturing
Seki Aoi Techno Co. Ltd, founded in Tokyo in 1969, boasts a rich legacy in manufacturing excellence for renowned Japanese companies spanning the Medical, Infrastructure and Printing industries. With decades of experience, we have honed our expertise to deliver unparalleled quality in medical equipment manufacturing. Operating from our cutting-edge facilities in Japan, China, and Malaysia, we uphold the highest standards of Japanese craftsmanship while leveraging strategic global partnerships to meet evolving industry demands.
Company Summary
Company Name |
Seki Aoi Techno Co., Ltd. |
|---|---|
Representative |
President and Representative Director: Mr. Tetsuo Seki |
Capital |
100 million Yen (=660,000 Euro) |
| Location | Headquarter: Tokyo, JAPAN |
| Established | May 1969 |
Our History
Seki Aoi Techno Co. Ltd. emerged in 2009 through the merger of Seki Electronics Ltd. (founded in Tokyo in 1969) and Aoi Camera (established in 1968). This union brought together the expertise of Seki Electronics in color graphics controllers, ozone meters, and photo composition systems with the manufacturing acumen of Aoi Camera deeply tied to Minolta, a prominent player in Japan's photography industry. Initially dedicated to commercial camera assembly, Aoi Camera swiftly transitioned into a leading manufacturer of Printed Circuit Boards and ventured into Medical Device production in 1996.
In 2010, under the umbrella of Seki Aoi Techno Co. Ltd., the company expanded its reach by establishing SEKI AOI TECHNO(WUXI)CO.,LTD., strategically located near Shanghai, one epicenter of China’s medical device manufacturing. This move aimed to capitalize on the burgeoning opportunities in China's medical device market. Subsequently, in 2015, SEKI AOI TECHNO(MALAYSIA)SDN.BHD. was inaugurated, further reinforcing the company’s broad Asian presence and commitment to delivering innovative solutions across borders.
Our Vision
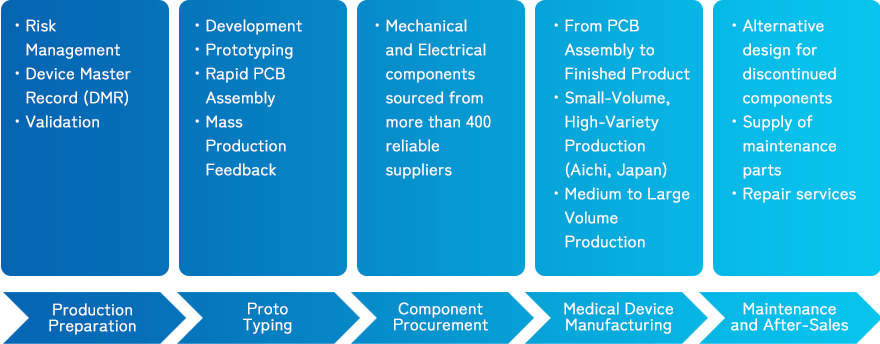
We want to be a leading One-Stop Service from the complete Medical Device Production for our partners from around Asia. From Prototyping, Production Preparation, Medical Device Manufacturing, Component Procurement as well as Maintenance and After-Sales.
■One-Stop Service for the complete Medical Device Production
Our Products
■Outsourced Development and Manufacturing (ODM) Achievements
- Image Synthesis Processing
- Silver Halide Photographic Development
- High-Speed Perforation Device for Copy Paper
- Blood Analysis Device
- Specimen Analysis Device
- Vision Measurement Device
- Urine Analysis Device
- Surgical Ultrasonic Unit, etc.
■Contract Manufacturing and Assembly (EMS) Achievements

- Jaundice Meter
- Luminance Measurement Device
- Color Difference Meter
- Ultrasonic Diagnostic Device
- Bone Densitometer
- Barcode Reader, etc.
Our Production facilities
We operate production facilities in three countries: Japan, China, and Malaysia, boasting a total workforce of over well-skilled 650 employees.
■JAPAN
| Country |
 |
|---|---|
| Company Name | SekiAoi Techno Co. Ltd. |
| Established | May 1969 |
| Functions | Centralized Operations of Overseas Production (Mother Factory) Parts Procurement, PCB Assembly, Prototyping, Mass Production of Finished Products |
| Certifications | ISO9001 / ISO14001 / ISO13485 - Medical Device Manufacturing Registration |
■CHINA
| Country |
 |
|---|---|
| Company Name | SEKI AOI TECHNO(WUXI)CO.,LTD. |
| Established | September 2010 |
| Functions | Parts Procurement, PCB Assembly, Prototyping, Mass Production of Finished Products |
| Certifications | ISO9001 / ISO14001 / ISO13485 - Foreign Manufacturing Establishment Registration |
■MALAYSIA
| Country |
 |
|---|---|
| Company Name | SEKI AOI TECHNO(MALAYSIA)SDN.BHD. |
| Established | May 2015 |
| Functions | PCB Assembly |
| Certifications | ISO9001 / ISO14001 |
Certifications
Our operations are proudly certified with ISO9001 / ISO14001 / ISO13485.

Navigating Stricter Regulations in China
In response to stricter regulations in China mandating a certain percentage of medical equipment manufacturing to be localized, Seki Aoi Techno Co., Ltd. stands as your ideal partner. With our high-quality manufacturing facilities in Wuxi, China, we ensure compliance with these regulations while maintaining our hallmark standards of precision and excellence. Moreover, our extensive network of local suppliers in China allows us to streamline production processes and access essential resources efficiently, providing a competitive edge in this dynamic market landscape.
Why Choose Seki Aoi Techno?
- 1.)Japanese Precision:
- Our commitment to precision and quality is ingrained in every aspect of our manufacturing process, ensuring reliable and innovative solutions for the medical industry.
- 2.)Global Footprint:
- With manufacturing facilities strategically located in Japan, China, and Malaysia, we offer flexibility and scalability to meet diverse market needs.
- 3.)End-to-End Solutions:
- From conceptualization to production, Seki Aoi Techno provides comprehensive manufacturing solutions tailored to the unique requirements of our partners.
- 4.)Compliance and Regulation:
- Stay ahead of regulatory requirements with Seki Aoi Techno’s thorough understanding of global standards and regulations governing medical equipment manufacturing.
- 5.)Technological Innovation:
- Through our advanced technical capabilities, we relentlessly pursue product performance and efficiency, ensuring the realization of our clients' productization goals through ongoing innovation.
- 6.)Supply Chain Excellence:
- Benefit from our robust supply chain management capabilities, ensuring seamless procurement and timely delivery of components and finished products.
- 7.)Collaborative Partnership:
- At Seki Aoi Techno, we prioritize collaborative partnerships, working closely with our clients to understand their needs and exceed expectations.
- 8.)Sustainable Practices:
- Committed to sustainability, we integrate eco-friendly practices into our manufacturing processes, minimizing environmental impact without compromising quality.
- 9.)Cost-Effective Solutions:
- Maximize cost efficiency without sacrificing quality with Seki Aoi Techno optimized manufacturing processes and value-driven approach.
Contact Us
We are interested in long-lasting partnerships with Medical Device manufacturers from around the Globe. Please get in contact with us:
